Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Comparative Study of Starch-Based Bioplastic from Banana (Musa acuminata) peel (SBB) Incorporated with Commercialized and Extracted Chitin
Authors: Mark Khevin L. De Guzman, April Jieren R. Muska
DOI Link: https://doi.org/10.22214/ijraset.2023.55639
Certificate: View Certificate
Abstract
The study produced extracted Chitin from Asian Green Mussel (Perna viridis) and Golden Apple Snail (Pomacea canaliculate) shells using two-way chemical extraction process; deproteinization and demineralization. The extracted and commercialized Chitin were characterized through Fourier Transform Infrared (FTIR) Spectroscopy to determine and compare the functional groups present. Starch-based bioplastics were produced using banana peel starch (BPS) incorporated with commercialized chitin and chosen chitin extract which is from AGM. The bioplastics were tested for its morphology through Scanning Electron Microscopy (SEM), physical property through air test, chemical properties through biodegradability test, chemical reactivity test, water vapor transmission rate (WVTR) test, water absorption test and flammability test, and mechanical property through tensile strength test. Four treatments were carried out using 1%, 2%, 3%, and 4% by weight chitin addition. The control SBB does not contain any chitin. All treatments were prepared using constant amount of BPS, PVA solution and glycerol. Based from the results, chitin addition improves the strong acid resistance, water-barrier properties, and tensile strength of the SBB. It was found out that the best treatment was T3 of AGM which has a WVTR of 73.9868 g/m2, dissolved in water in an average of 6.1783 days, and has a tensile strength of 64286.3777 Pa.
Introduction
I. INTRODUCTION
A. Background of the Study
The petrochemical industry paved the way for the birth of plastics, a material which is now widely used for single-use packaging. Manufacturers consider plastics ideal for packaging because of its lightweightness, durability, flexibility, and water resistance. According to the British Plastics Federation (2022), this polymeric material requires cheap raw materials and less energy to transport and mass produce than other packaging, thus making it more cost-effective.
However, plastics are not able to break down easily and it takes about 20-500 years for it to completely decompose, depending on the material and structure (Armentrout, 2021). The rapid increase in production and excessive usage of single- use plastics resulted in massive plastic pollution, one of the environmental concerns that human struggles to battle up to this day. Based on a study published in Science Advances journal, 6.3 billion of the 8.3 billion metric tons that has been totally produced in the last six decades has become plastic waste (Parker, 2019). It is alarming that the recycling rate of plastics in 2018 is only 8.7 percent resulting from it taking over landfills and deep oceans. In fact, it was discovered that plastic accumulation already makes up about 60%-80% of all marine debris studied (Rios et al., 2007).
It is impossible to eradicate the influence and utilization of plastics despite its negative impact to the environment because this material is not only used for mere convenience. Fortunately, the emergence, development, and commercialization of biodegradable plastics can mitigate the effects of plastic pollution. Biodegradable plastic is derived from plant-based material that can be broken down by microbes into water, carbon dioxide or methane, and compost under certain conditions. Approximately 50% of the bioplastics widely used for packaging applications are prepared from starch (Gadhave et al., 2018). The only downside of starch-based bioplastics is its hydrophilic properties, it cannot withstand humidity nor oppose water. Based on research, blending starch-based bioplastics to chitin, a biopolymer with hydrophobic properties can reinforce its water resistance properties.
Chitin is considered to be the most abundant amino polysaccharide occurring in nature, and waste from aquatic species are its well-known alternative resources.
The shell of selected crustacean was reported to consist of 30-40% protein, 30-50% calcium carbonate and calcium phosphate, and 20-30% chitin (Fernandez, 2004, as cited in Abdulkarim et al., 2013).
In connection, this study was conducted to replicate the same success of bioplastic packaging from shrimp shells using Asian green mussel (AGM) as an additive to banana peel starch. The said raw material for chitin-extraction is considered to be invasive as well as an abundant food waste in the South-East Asian region. It is estimated that more than 250,000 metric ton of shell, scale, and muss carapace waste are produced in the Philippines (Cadano et al., 2020). Hence, conducting this study will not only solve the overdue problem in plastic pollution but will also help in the food waste management, create a sustainable industry, and promote a balanced ecosystem.
B. Objectives of the Study
This study generally aimed to compare the effects of commercialized chitin (CC) and chosen chitin extract (CCE) from either Asian Green Mussel (AGM) and Golden Apple Snail shells (GASS) on the properties of SBB produced.
Specifically, the study aimed to:
- Extract and characterize the chitin extracted from AGM and GASS through Fourier Transform Infrared (FTIR) Spectroscopy and compare with the CC;
- Identify which extract is to be used for the production of SBB;
- Optimize the amounts of chitin from CC and CCE to produce SBB;
- Characterize the properties of the SBB through
a. surface morphology using Scanning Electron Microscopy (SEM) analysis;
b. physical property test;
c. chemical properties test;
- biodegradability test,
- strong and weak acid test,
- strong and weak base test,
- organic solvent test,
- water vapor transmission rate and absorption test,
- flammability test
d. mechanical property test;
5. Compare the properties of the SBB incorporated with CC and CCE with the control SBB; and
6. Identify which SBB incorporated with CC and CCE will give a better yield as a bioplastic in terms of physical property, chemical properties, and mechanical property as compared with the control SBB.
C. Significance of the Study
The findings of this study under proper analysis and guidance will benefit the environment by mitigating plastic pollution. Biodegradable plastic can lessen the carbon footprint coming from the production of petroleum-based plastics, and reduce the non-biodegradable wastes piled in landfills (Atiwesh et al., 2021).
Due to environmental implications of the use of plastics, the use of bioplastics has gained interest to many. This study will greatly impact society as well by converting food waste into a useful material. This will provide opportunities to fisherfolks and business owners to maximize their income by selling or utilizing the materials instead of discarding them to the environment.
Moreover, this study will also be beneficial to commercial industries by providing an alternative way for a sustainable and biodegradable packaging which is rapidly gaining popularity in the market. As compared to conventional plastics, bioplastics are cost and energy efficient during production and they do not contain health damaging additives like phthalates and bisphenol-A (Cho, 2017).
Lastly, it will benefit future researchers who will conduct a study related to chitin extraction and production of bio-plastic. This can serve as a reference which in turn can lead to further development of the product.
D. Scope and Limitations of the Study
The study compared the effects of chitin addition from CC and CCE on the properties of SBB, which was carried out with four treatments using 1%, 2%, 3%, and 4% by weight chitin addition, respectively. The CCE used in the study was chitin extracted from AGM because of its higher percentage of recovery compared to that of the GASS. The chitin samples were characterize using Perkin Elmer FTIR Spectrometer Frontier with a wavelength spectrum between 600 and 4000 cm-1.
The control SBB does not contain any chitin. The amounts of 20 grams of starch from banana peel, 100 mL of 0.465 M polyvinyl alcohol (PVA) solution, 2.5 mL of glycerol as plasticizer, and 10 mL of distilled water were maintained in all the treatments (Cabacungan, 2006).
The produced SBB was characterized in terms of physical, chemical, and mechanical properties. For the physical property, inside and outside air tests were employed. For the chemical property, samples were subjected to strong and weak acids, strong and weak bases, and organic solvent tests to determine their chemical reactivity. Water vapor transmission rate and absorption tests were also employed to determine its water-barrier properties. Horizontal and vertical flamability tests were also conducted. Moreover, the samples were analyzed using SEM after four weeks of a degradation period in a controlled compost. For the mechanical property, tensile strength was calculated.
II. REVIEW OF RELATED LITERATURE
???????A. Plastics
Conventional plastics are materials belonging to the wide range of synthetic or semi-synthetic organic solids that are most commonly derived from petrochemicals. The global petroleum-based plastic production in 2019 totalled around 368 million tons which is far-off from where it started in 1950 with just 1.7 million tons (Tiseo, 2021). The urbanization during the twentieth century is said to be the cause of heightened demand and utilization of petroleum-based plastic materials in various applications, especially in the medicinal, industrial area, commercial, and municipal field. This material is used in an enormous and expanding range of products and applications due to its relatively low cost, ease of manufacture, versatility, and imperviousness to water.
In connection with the mentioned characteristics, petroleum-based plastics became the most popular and suitable material for disposable and single-used packaging. According to the National Geographic Society, about 40 percent of plastics produced is for packaging that is used only once and then discarded. Petroleum-based plastics may be fragmented but cannot naturally break down due to their profound stability. Stabilizers and plasticizers that are added to enhance the characteristics and efficiency of petroleum-based plastic materials can be leaked into the environmental system during the degradation process (Suman et al., 2020). The material decomposition time is significant and consequently, making most conventional plastics waste such as polyethylene, polypropylene, polystyrene, polyvinyl chloride, polyurethane and polyethylene terephthalate accumulate in the environment. Studies also showed that the ingestion of micro-plastics and their by- products (BPA and DEHP) can trigger health problems.
Plastic pollution has been recognized as one of the most critical environmental challenges, affecting all life forms, ecosystems and economy, worldwide. The Philippines alone generates 2.7 million tons of plastic waste annually, 20 percent of which is estimated to end up in the ocean. According to Global Alliance for Incinerator Alternatives (GAIA) (2020), the country uses almost 60 billion sachets a year; with 0.28-0.75 million tons of plastics entering from coastal area it makes it one of the world’s worst offenders on marine pollution.
The Philippines also dominated the global top ten list of largest plastic emitting rivers by having seven placements contributing about 1/3 of the global total. Ranking first is the Pasig River with its 6.43% share of global ocean plastic pollution, even higher than the contribution of North America (5.4%), Europe (0.6%), and Oceania (037%) combined (Meijer et al., 2021).
- Biodegradable Plastics
The initiative to lessen the harmful effects of conventional plastics in the environment gave birth to the innovation of biodegradable plastics. Bioplastics are used as an alternative to conventional fossil fuel based plastics and are increasingly being used in food contact materials (Wagner, 2014). These are plastics that are made from plants and/or other biological materials such as vegetable fats and oils, corn starch, straw, wood chips, food waste, agricultural by-products rather than petrochemical materials. It is either biobased, biodegradable, or features both properties. Bioplastic is characterized as transparent, flexible, durable, and a heat resistant material. There are three types of bioplastics: Starch-based bioplastics, Cellulose-based bioplastics and Protein-based bioplastics. The bioplastics most commonly used in packaging includes starch-based blends, polylactic acid-based blends (PLA blends), thermo-plastically modified starch (TPS), cellulose-based plastics, and biobased bioplastics (bioPET).
Bioplastic materials from renewable polymers, like proteins, constitute a highly interesting field for important industrial applications such as packaging, agriculture, etc., in which thermo-mechanical techniques are increasingly being used. The primary benefit and advantage in using bioplastics is their capability to improve the environmental impact of a product. It uses renewable resources for monomers production which reduces the use of fossil fuels and greenhouse gas emissions.
The biodegradable nature of bioplastics offers an additional option for the end-of-life of the products, which allows for a reduction in the amount of waste material. However, degradable plastic materials can still last for many years, depending on environmental circumstances, such as temperature, oxygen, and the exposure levels of ultraviolet (UV) light. The required characteristics of the bioplastics are achieved by fine-tuning the quantities of the additives.
???????2. Starch-based bioplastics
Starch-based bioplastics are derived from starchy raw materials and are often mixed with biodegradable polyesters. Since starch is totally biodegradable, it can be hydrolyzed into glucose by microorganisms or enzymes which metabolize it into carbon dioxide and water. Due to the abundance and simple production of starch- based bioplastic, it is widely used for packaging applications. Researches have shown that approximately 50% of the bioplastics used commercially are starch-based. It holds many advantages such as being eco-friendly, renewed easily, biocompatible with the human body, non-toxic and simply degraded in a short time. The tensile properties of starch are suitable for the production of packing materials, for trade applications, tit is regularly mixed with eco-friendly polyesters. (Marichelvam et al., 2019).
Unfortunately, because of the starch-based bioplastics’ hydrophilic properties, it exhibits low mechanical properties and cannot resist humidity. To reinforce its water barrier properties, starch based bioplastics were blended with synthetic polymers such as PE, LDPE, HDPE, etc. Nevertheless, the synthetic polymer reduces biodegradability of starch based bioplastics and it is not compatible with the environment. Furthermore, safety will always be considered as a priority regarding any additives for food packaging applications. Based on this concern, natural filler and edible reinforce agents, such as natural fibers, starch or cellulose crystals, and laver have been used to improve starch-based materials (Jiang et al. 2019). One of biopolymers with hydrophobic properties that can be used to optimize water resistance of starch-based bioplastics is chitin. Chitin develops advantages such as stable to chemical reactions, low chemical selectivity, non-toxic, biodegradable and insoluble in water (Abdullah et al., 2020).
B. Banana Peel
Saba bananas, botanically classified as Musa paradisiaca or Musa acuminata x balbisiana, is a locally-grown fruit in the Philippines that is available in the market year-round. It is a very abundant agricultural produce such that in 2019, the Philippines ranked second in terms of world export, next to Ecuador. The said banana cultivar has a very high starch content approximately 68.1%, that is way higher than other tropical banana cultivars (Hongpattarakere and Uraipan, 2014).
Banana peels are richer in phytochemical compounds than the pulp. It has also anti-fungal, antibiotic properties which can be beneficial to human health and are packed with polyphenols, carotenoids, and other antioxidants that fight cancer- causing free radicals in your body.
According to the most recent Philippine survey, 25% of the country’s net banana production, or around 2 million metric tons for 2019, undergoes food processing. The preliminary studies showed that after the processing, the waste produced is around two-thirds of the fresh material in terms of weight. Hence, the potential waste generation from banana processing is more than a million metric tons (Rivadeneira et al., 2020). Moreover, it is estimated that over 114.08 million metric tons of banana peels are thrown away worldwide annually, leading to disposal by burning which is environmentally unfriendly (Huzaisham and Marsi, 2020). Utilizing banana peel as a raw material for the production of biodegradable plastics will not only mitigate the effects of plastic pollution but will also lessen food wastes.
???????C. Glycerol
The most commonly used plasticizer to produce starch-based biodegradable films is glycerol. It is an alditol and a triol with a structure of propane substituted at positions 1, 2 and 3 by hydroxyl groups. Starch and glycerol melt and flow at temperatures between 90°C and 180°C under shear stress, producing thermoplastic starch allowing the use of injection, extrusion and blowing equipment. Plasticizers create greater flexibility in the polymer structure by reducing the intermolecular forces and the glass transition temperature of the material. This increases the mobility of the polymer chains in the starch films. The required proportion of starch and glycerol depends on the type of starch used (Blick et al., 2015 as cited in Marichelvam, et al., 2019).
???????D. Polyvinyl Alcohol
Polyvinyl alcohols (PVA) are synthetic polymers used since the early 1930s in a wide range of industrial, commercial, medical and food applications including resins, lacquers, surgical threads and food-contact applications. Addition of polyvinyl alcohol (PVA) has been shown to successfully reduce the brittleness of starch bioplastic (Judawisastra et al. 2017). Polyvinyl alcohol is a water-soluble and biodegradable polymer that possesses high bio-compatibility, but, more interestingly, and is capable of self-cross-linking due to the high density of hydroxyl groups located on its side chains. Conversely, PVA also undergoes fast hydrolysis and is bio-inert; thus, protein and cell adhesion are limited on the pure material and is mainly used to manufacture water-soluble films (Havstad, 2020).
???????E. Chitin
Chitin (β-(1–4)-poly-N-acetyl-D-glucosamine) is widely distributed in nature, it is the second most abundant polysaccharide after cellulose and is the most abundant amino polysaccharide polymer occurring. It behaves similarly to keratin and is the building material that gives strength to the exoskeletons of crustaceans, insects, and the cell walls of fungi (Rinaudo, 2006 as cited in Abdulkarim et al., 2013).
The production of chitin can be done in two ways, biological and chemical processes. Chitin can be extracted from living organisms in the lower plant and animal kingdoms, serving in many functions where reinforcement and strength are required. (Elieh-Ali-Komi and Hamblin, 2016).
Further, chitin develops advantages such as stable to chemical reactions, low chemical selectivity, non-toxic, biodegradable and insoluble in water. However, chitin structure is fragile and should be mixed with other materials to generate bioplastics (Oyekunle and Omoleye, 2019).
???????F. Extraction of Chitin
- Deproteinization
The deproteinization requires the disruption of the chitin protein-complex and the solubilization of proteins. This is performed by using chemicals that also depolymerize the biopolymer. The complete removal of protein is especially essential for biomedical applications, as a percentage of the human population is allergic to shellfish due to its protein component.
According to Younes and Rinaundo (2015), chemical methods were the first approach used in deproteinization. A wide range of chemicals including NaOH, Na2CO3, NaHCO3, KOH, K2CO3, Ca(OH)2, Na2SO3, NaHSO3, CaHSO3, Na3PO4 and Na2S. In addition of deproteinization, the use of NaOH invariably results in partial deacetylation of chitin and hydrolysis of the biopolymer lowering its molecular weight.
2. Demineralization
Demineralization is an important step in the chitin purification process; it is concerned in the removal of minerals, primarily calcium carbonate, using hydrochloric acid as a preferred reagent. It can be easily achieved because it involves the decomposition of calcium carbonate into the water-soluble calcium salts with the release of carbon dioxide as shown in the following equation:
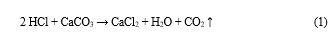
Most of the other minerals present in the shellfish cuticle react similarly and give soluble salts in presence of acid. Then, salts can be easily separated by filtration of the chitin solid phase followed by washing using deionized water (Percot et al., 2003 as cited in Younes and Rinaundo, 2015).
G. Asian Green Mussel
The Asian Green Mussel with a scientific name Perna viridis has been recognized as an invasive species since its introduction out of its native range around the world. It has the ability to form dense populations, up to 35,000 shells per square meter, on a variety of structures including vessels, wharves, mariculture equipment, buoys and other hard substrata (Benson et al., 2001). Because of its dispersed spawning nature, lack of local predators, fast growth, and high tolerance of environmental conditions, this mussel population is expected to expand in Atlantic habitats until it reaches its thermal limits (De Victor and Knott, 2005).
Generally, mussel shells consist of 30-40% protein, 30-50% calcium carbonate and calcium phosphate, and 20-30% chitin (Fernandez-Kim, 2004). Depending on the processing method used to derive the chitin (Khora and Limb, 2003), and also depending on the source, its degree of deacetylation may range from 30% to 95% (Martino et al., 2005 as cited in Abdulkarim et al., 2013). The very short food chain, sturdy nature, fast growth rate, and rare occurrence of catastrophic mass mortality caused by parasitic microorganisms of asian green mussel makes it possible to produce large quantities at a very reasonable price.

I. Characterization of Chitin
- FTIR (Fourier Transform Infrared Spectroscopy)
Fourier Transform Infrared Spectroscopy (FTIR) is attractive due to its nondestructive character, fastness, sensitivity, and suitability for both soluble and insoluble samples (Boukhlifi, 2019).
In infrared spectroscopy, radiation in different wavelengths is released onto the sample where a certain degree of radiation will be absorbed by the sample. This will then form a spectrum showing the absorption and transmission of the sample molecule which correspond to the frequencies of vibrations between the bonds of the atoms which compile the material (Kaewboonruang et al., 2016). Moreover, FTIR analysis is also commonly used to identify various functional groups constituting a compound. It is a rapid and non-destructive technique for the qualitative and quantitative determination of biomass components in the mid-IR region providing information about molecular fragments, the presence or absence of specific functional groups and can give an even deeper insight into the bioplastics’ structure (Hospodarova, 2018).
2. SEM (Scanning Electron Microscopy)
Scanning Electron Microscopy is a test process that scans a sample with an electron beam to produce a magnified image for analysis. The signals generated during SEM analysis produce a two- dimensional image and reveal information about the sample, including external morphology (texture), chemical composition, when used with the EDS feature, and orientation of materials making up the sample (Swapp, 2017). This technique allows examining the changes in the morphology of materials at the micro scale.
3. ISO 20200:2015 Biodegradability or Disintegration Test
Biodegradation is a process by which microbial organisms transform or alter the structure of chemicals introduced into the environment. Composting also plays an important role in biodegradability test. The optimum Carbon to Nitrogen ratio (C:N) to begin composting is 30:1. If the ratio increases, the decomposition is slowed; if the ratio decreases foul odors and nitrogen loss can occur. Food waste is typically 15:1, fruit waste 35:1, leaves 60:1, bark 100:1, and sawdust 500:1 (Risse and Faucette, 2017 as cited in Palaniveloo, K. et al. 2020).
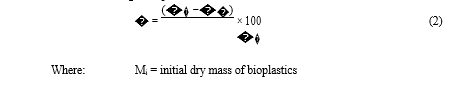
ISO 20200:2015 specifies a method of determining the degree of disintegration of plastic materials when exposed to a laboratory-scale composting environment. The degree of disintegration is generally measured in accordance with the percentage of particles which are retained on a sieve of 2 mm. It should be noted that the equation substantially represents the loss of mass in the conditions set by the test and can be calculated using the equation:
4. ASTM D543 Evaluating the Resistance of Plastics to Chemical Reagents
Chemical resistance testing is carried out by immersing samples in strong and weak acid, strong and weak base, and organic solvent. The type of chemical reagents to be used are specified in applicable standards to reflect the intended service conditions. Plastics are susceptible to crazing or cracking following exposure to certain reagents.This test covers the evaluation of plastic materials for resistance to chemical reagents, simulating performance in potential end use environments (https://www.astm.org/d0543-21.html).
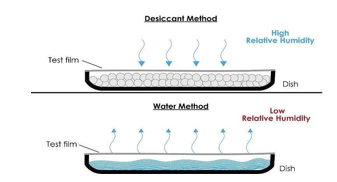
5. ASTM E96 Water vapor transmission rate (WVTR)
Water vapor transmission rate is the standard measurement by which films are compared for their ability to resist moisture transmission, with lower values indicating better moisture protection. Only values reported at the same temperature and humidity can be compared, because transmission rates are directly affected by both of these parameters. (https://www.astm.org/e0096-e0096m-22.html).
6. Water Absorption Test
Water absorption of polymer blends is one of the important properties for evaluating their stability on water affecting their long- term performance. The test was to identify the time needed for the bioplastic to dissolve in water. Using water as the solvent, the bioplastics are immersed in the container and the time duration for the complete dissolution is recorded.
7. UL94 Flammability Test
Flammability test determines how a material will ignite or burn when exposed to or used near fire or heat. The Underwriters Laboratory (UL94) is the most widely used standard to evaluate the flame retardant performance of plastic materials. The said testing is the first step toward obtaining a plastic recognition and subsequent listing in the Plastics Recognized Component Directory (https://omnexus.specialchem.com/).
8. Horizontal Burning
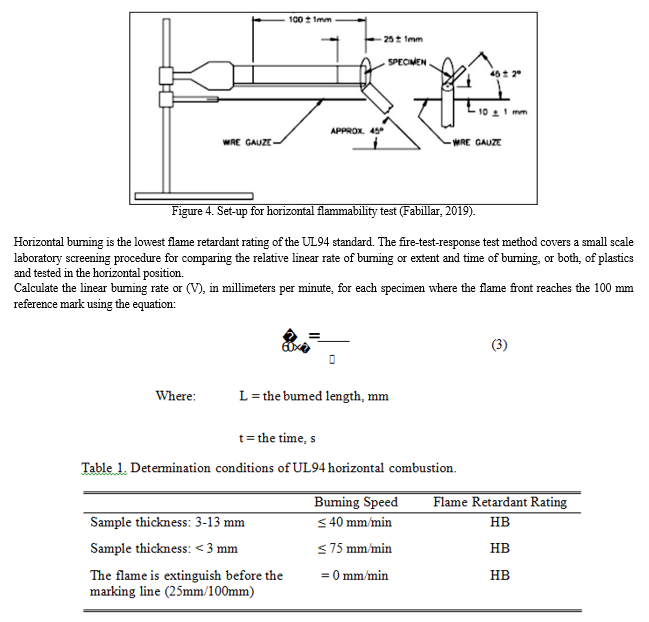
9. Vertical Burning
This test measures the self-extinguishing time of the vertically oriented polymer specimen. The test evaluates both the burning and afterglow times and dripping of the burning test specimen. The material will be rated by V-0 if the flame extinguishes within 10 seconds with no dripping, V-1 if the flame extinguishes within 30 seconds with no dripping, and V-2 if flame extinguishes within 10 seconds with dripping.

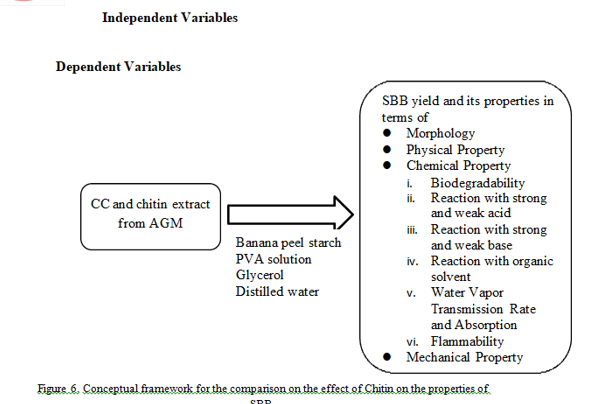
III. METHODOLOGY
A. Locale of the Study
The extraction of chitin as well as the production, testing, and characterization of the biodegradable plastic were conducted at the laboratory of Department of Chemical Engineering, Mariano Marcos State University, City of Batac, Ilocos Norte. The raw materials were collected from public markets and residential waste for saba banana peels, food wastes from seafood restaurants and residential for asian green mussel shells, and rice fields in the Brgy. 8, San Antonio, Sarrat, Ilocos Norte for the golden apple snail shells. The samples for Fourier Transform Infrared Spectroscopy (FTIR) were sent to Advanced Device and Materials Testing Laboratory (ADMATEL) and samples for Scanning Electron Microscopy (SEM) were sent to Center for Innovative Materials in Emerging Applications (CIMEA) for characterization.
B. Research Design
The research design that was used in the study is a combination of experimental, comparative and descriptive methods. The experimental design was employed in the extraction of chitin and production of SBBs. The comparative and descriptive designs were used to characterize the properties of the SBB produced. Moreover, Analysis of Variance (ANOVA) was employed to determine the best parameters as it analyzes factors that affect the given data set.
C. Research Materials
- Materials
Saba Banana Peel Hydrochloric acid (HCl), 37% (Emsure ACS) Asian Green Mussel Shell Sodium metabisulfite (Na2S2O5), (Dalkem) Golden Apple Snail Shell Sodium hydroxide (NaOH), (HIMEDIA)
Commercialized Chitin (CC), (HIMEDIA)
Ammonium hydroxide (NH4OH), (HIMEDIA)
Glycerol (C3H8O3), (TELSTAR) Sulfuric acid (H2SO4), 98% (ACILabscan) Polyvinyl Alcohol (PVA), (Dalkem) Acetic acid (CH3COOH)
Distilled water Ethanol (C2H5OH), (DKL)
2. Laboratory Apparatus and Equipment
Mortar and Pestle/ Blender Aluminum tray
Beaker Funnel and filter paper
Petridish Pipette and aspirator
Strainer Spring balance and weights
Oven (Binder ED53) pH meter and litmus paper
Analytical Balance (Ohaus PA413 and Radwag AS220)
Hot Plate Stirrer (Corning PC-620D, Corning PC-420D and Witeg SMHS-3)
Perkin Elmer FTIR Spectrometer Frontier
Scanning Electron Microscope (Hitachi FlexSEM 1000 Variable Pressure)
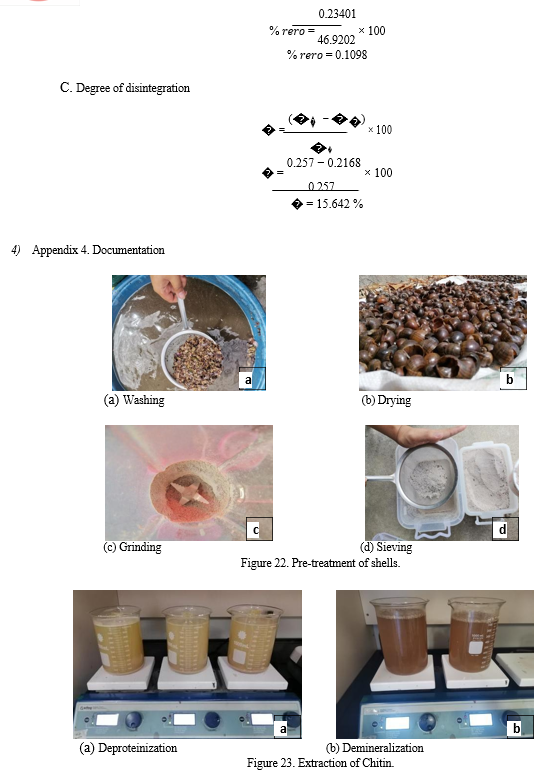
D. Research Procedure
- Extraction of Starch from Banana Peel
The banana peels were cut into small sizes and dipped in 0.5 % wt Na2S2O5 solution prior to the boiling process. An 800 ml beaker was filled with distilled water and heated over a hot plate where the banana peels were boiled for 30 minutes. After the boiling process, the water was decanted off and the banana peels were placed in an aluminum tray and covered with a dry gauze pad prior for air-drying. The peels were pureed using a blender and filtered to produce banana peel starch (BPS).
2. Extraction of Chitin
Deproteinization: The shells obtained were scraped free of loose tissue, washed thoroughly with water, and dried in an oven to constant weight. The dried shells were size-reduced using a blender. The powderized shells were deproteinized using 2M NaOH with a ratio of 20 ml : 1 g (v/w) of NaOH to shell by stirring with a mechanical stirrer at a speed of 150 rpm. This was done at room temperature for the duration of 2 hours. The mixture was washed with distilled water, filtered and tested for its pH. These steps were repeated until the pH is neutral. The deproteinized shells were then dried in the oven at 110°C until constant weight was obtained.
Demineralization: The residue from the deproteinization process was weighed. It was treated with 2 M HCl with a ratio of 20 ml : 1g (v/w) by stirring with a mechanical stirrer at a speed of 150 rpm. This was done at room temperature for the duration of 2 hours. These steps were repeated until the pH is neutral. The extracted chitin were then dried in the oven at 110°C until constant weight was obtained.
Chitin extracted from AGM and GASS and the procured CC were characterized by FTIR-ATR Analysis. Test results were obtained using the Perkin Elmer FTIR Spectrometer Frontier of wavelength spectrum between 600 to 4000 cm-1. The extract that contained the highest amount of chitin was chosen and used in the preparation of SBB. In this case, AGM yield a higher amount of chitin than GASS.
3. Preparation of Controlled Compost
Controlled compost was prepared in a 60L rectangular prism container prior to the biodegradability test. The first layer consisted of 6-8 inches of brown materials composed of dried leaves, twigs and shredded paper and green materials composed of kitchen waste and grass clippings. The layer was topped with 1-2 inches of chicken manure to provide nitrogen for the bacteria and microorganisms. The top layer contained 1-2 inches of garden soil.
4. Preparation of PVA solution
Four grams of polyvinyl alcohol powder was diluted in 100 mL of distilled water in a 150 mL beaker. The solution was heated on a hot plate at 100°C with continuous stirring until a homogeneous mixture was obtained.
5. Optimization of chitin extract
The optimization of the chitin extract from AGM and CC was carried out using 4 treatments. In all the treatments, constant amounts of banana peel starch, polyvinyl alcohol (PVA) solution, glycerol, and distilled water were used. The amounts of chitin were 1.0 wt% treatment 1 (T1), 2.0 wt% for treatment 2 (T2), 3.0 wt% for treatment 3 (T3), and 4.0 wt% for treatment 4 (T4) (Abdullah et al., 2020). Triplicates were done for all the treatments.
6. Production of control SBB
Production of control SBB was done by combining 20 grams of BPS, 100 mL of PVA solution, 2.5 mL of glycerol, and 10 mL of distilled water. The mixture was boiled to 80°C using a hotplate. When the mixture achieved a sticky paste consistency, it was poured and spread out to an aluminum tray. The tray was placed immediately into the preheated drying oven at 110oC for the duration of 15 minutes. The SBB was cooled before scraping the film off the surface.
7. Production of SBB using AGM chitin extract
Production of SBB was done by combining 20 grams of BPS, 100 mL of PVA solution, 2.5 mL of glycerol, 10 mL of distilled water with 1.0 wt% of extracted chitin from AGM. The mixture was boiled to 80°C using a hotplate. When the mixture achieved a sticky paste consistency, it was poured and spread out to an aluminum tray. The tray was placed immediately into the preheated drying oven at 110oC for the duration of 15 minutes. The SBB was cooled before scraping the film off the surface. This was treatment 1 (T1).
For the succeeding treatments (T2-T5), the above procedures were followed using varying amounts of 2.0 wt%, 3.0 wt%, and 4.0 wt% of chitin from AGM for T2, T3, and T4, respectively.
8. Production of SBB using CC
The same procedures in the production of SBB using AGM were employed in the production of SBB using CC, replacing only the AGM into CC.
9. Characterization of SBB
The SBB produced underwent several tests to determine its properties. The results of the tests consequently determined which treatment yielded the best results. Three replicates were done for each treatment.
a. Surface Morphology: The morphology of the sample was studied on a field emission scanning electron microscope. The samples were dried in the oven at 80 oC for the duration of 2 hours, sealed in a petri dish, and sent to CIMEA for characterization using Hitachi FlexSEM 1000 Variable Pressure at 10.0 µm and 500 µm magnification for surface and cross-sectional, respectively. The tests for physical and chemical properties were adopted from the research of Cabacungan (2006). The water vapor transmission rate and water absorption test were adopted from the research of Abdullah et al. (2020) while the flammability and mechanical tests were adopted from the research of Fabillar (2019). All tests were done in triplicates.
b. Physical Property Test: Air tests were used to determine the physical property of the bioplastics produced. The strips of the newly prepared film were exposed to open air, indoor and outdoor for one month. The changes in its appearance were recorded weekly.
c. Chemical Properties Test: Biodegradability test, strong and weak acid test, strong and weak base test, organic solvent test, water vapor transmission rate test, water absorption test and flammability test were used to determine the chemical properties of the bioplastic produced.
d. Biodegradability Test: Twelve strips per SBB sample were buried in a depth of 6 inches in the prepared compost for a period of 4 weeks at room temperature. The samples were taken out weekly to record its visual appearance. In the fourth week, the degree of disintegration was calculated using the initial and final mass of the retrieved samples. The morphology of the samples will also be recorded for comparison.
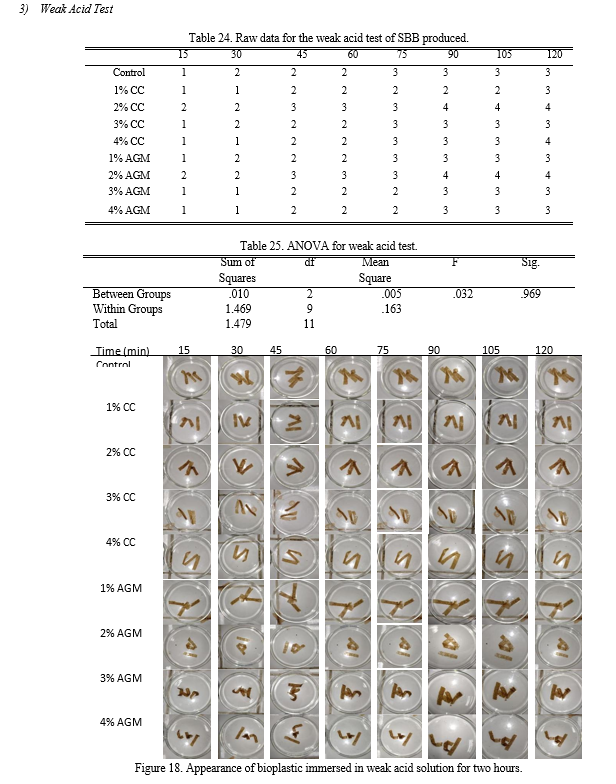
e. Strong and Weak Acid Test: The strips of the sample were immersed in eighteen (18) separate petri dishes, nine of which contained a strong acid (1M sulfuric acid), and the other nine contained a weak acid (1M acetic acid). The changes in its appearance were recorded every 15 minutes for two hours.
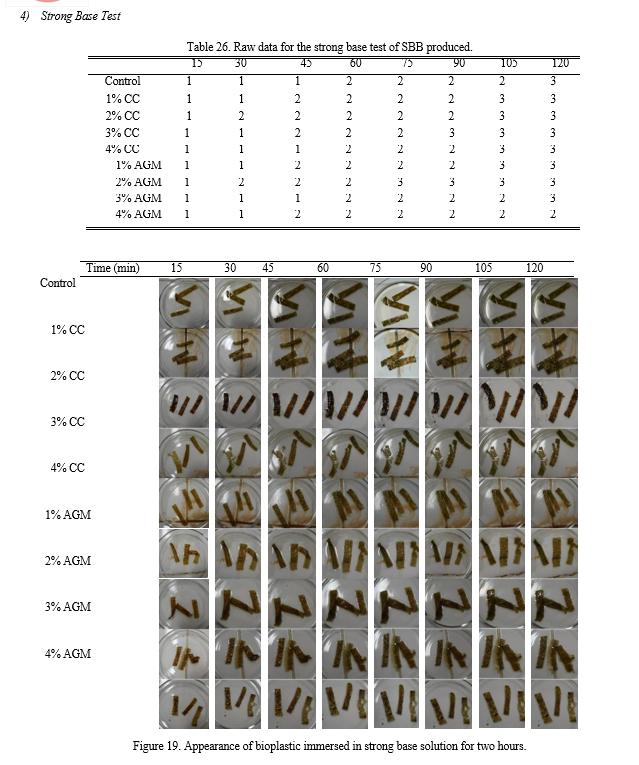
f. Strong and Weak Base Test: The strips of the sample were immersed in eighteen (18) separate petri dishes, nine of which contained a strong base (1M sodium hydroxide), and the other nine contained a weak base (1M ammonium hydroxide). The changes in its appearance were recorded every 15 minutes for two hours.
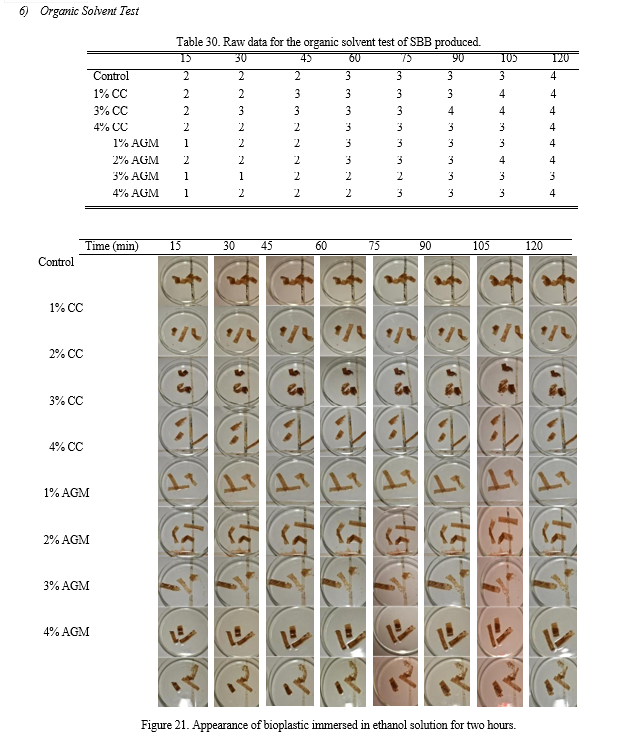
g. Organic Solvent Test: The strips of the sample were immersed in nine (9) separate petri dishes which contained a 20% (v/v) ethanol. The changes in its appearance were recorded every 15 minutes for two hours.
h. Water Vapor Transmission Rate Test: The bioplastic strips were sealed on holders which had been filled with water for 30 minutes. Samples were then placed in a desiccator by condition at relative humidity 50 % and temperature of ±25 °C and were evaluated every 2 hours for 8 hours to determine the transformation mass.
i. Water Absorption Test: The bioplastic strips were immersed in distilled water at room temperature. The changes were observed and the time for the sample to be completely dissolved was recorded.
j. Flammability Test (Horizontal Test): Samples with a volume of 500 mm3 were used in the test. The samples were attached to an iron stand clamped in a horizontal position and were subjected to flame through a candle. After ignition, the flame was removed from the samples and the times needed to completely burn the samples were recorded.
k. Flammability Test (Vertical Test): Samples with a volume of 500 mm3 were used in the test. The samples were attached to an iron stand clamped in a vertical position and were subjected to flame through a candle. Withdrawal of the flame was done after the ignition of the sample. The time when the first drip dropped from the samples was recorded.
l. Mechanical Property Test: Tensile stress test was used to determine the mechanical property of the bioplastics produced. The strips with an area of 0.5 in2 were hooked on a spring balance pulling it apart.

IV. RESULTS AND DISCUSSION
This study aimed to produce SBB incorporated with CC and CCE which is from AGM, specifically to optimize the amount of chitin and to identify which of the raw materials can give a better yield of bioplastic. The chitin samples used for the production were subjected to FTIR-ATR spectrophotometer to identify its functional groups.
Bioplastics were produced by mixing the desired proportions in each treatment and heating it at a temperature of 80ºC until a sticky glue-like consistency was attained. The mixture was then spread on a tray and put in a preheated oven at 110ºC for at least 2.5 hours. The samples were then cut into strips for testing.
A total of 459 sample strips were used for the different tests which included SEM analysis, air test, biodegradability test, chemical tests, water vapor transmission and absorption tests, flammability test, and tensile strength test. Three replicates were done for each treatment.
Analysis of Variance (ANOVA) was used to analyze the data gathered from the different tests conducted. It determined which treatment was the best for each raw material for bioplastic based from tests conducted. It was also used to determine which was the best bioplastic produced using all the treatments of both raw materials.
A. Extraction and Characterization of Chitin
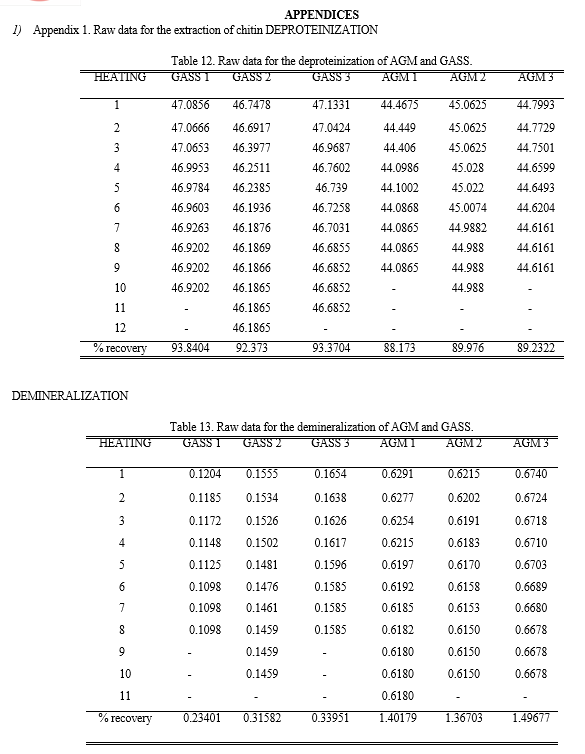
The extractions of chitin from the shells were divided into two main parts, deproteinization using 1M NaOH, and demineralization using 1M HCl. The percent recovery of deproteinize shell is higher in GASS however chitin recovered from the demineralization process is much lower compared to that of AGM.
These means that using GASS as a raw material for chitin extraction is not efficient and costly. There is a vast amount of raw materials as well as chemical for the extraction process needed to produce the amount required for the optimization. This is the reason why commercialize chitin labeled as technical grade chitin from shrimp shells was used for the comparison.
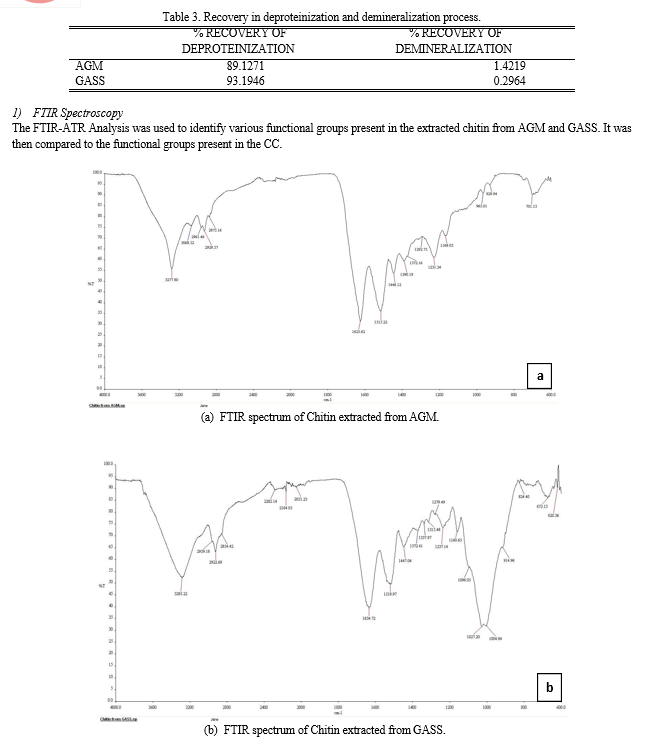
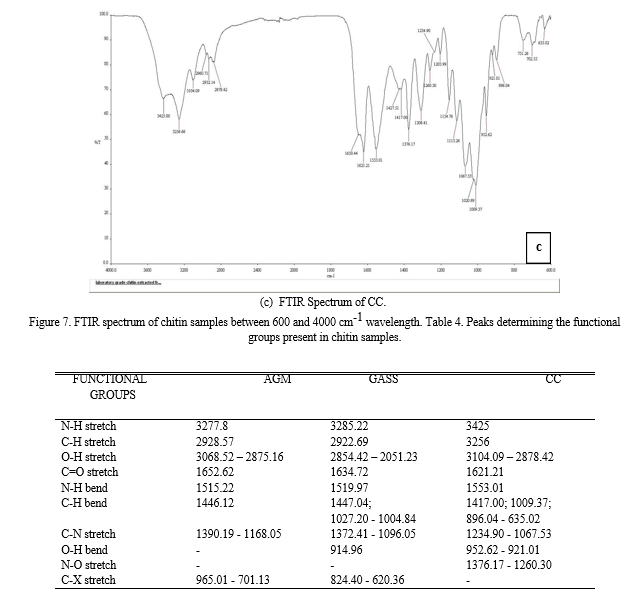
Based from the results of FTIR-ATR analysis, chitin contains Amino or Amide group (-NH2) from the stretching vibration and bending vibration of N-H; Alkyl group (-CH2- or –CH3) from the stretching vibration and bending vibration of C-H; Hydroxyl group (-OH) from the stretching vibration of O-H; and Carboxyl group (-COOH), Aldehyde group (-CHO) or ester function (-COOR) from the stretching vibration of C=O. In the fingerprint region (1500 cm-1 to 500 cm-1) where it contains the complex set of absorptions, it is found that all contains Aliphatic amines (C-N stretch). Both the AGM and GASS contains Alkyl Chloride while both the GASS and CC contains Carboxylic acid (O-H bend). The CC has unique functional group present in it which is the Nitro compound (-O-N=O).
B. Properties of starch-based Bioplastics
- Physical Property (Air Test)
The physical property of the bioplastics was tested by exposing them to indoor and outdoor open air. The changes in appearance and color were observed and recorded weekly for one month. Likert scale was used to quantify and characterize the appearance of the bioplastics. The changes in appearance were mostly documented early in the morning where the dampness of the air was higher compared to other time of the day. Since the polymer used is soluble in water, it can be a factor that has affected the bioplastics to react with moisture.
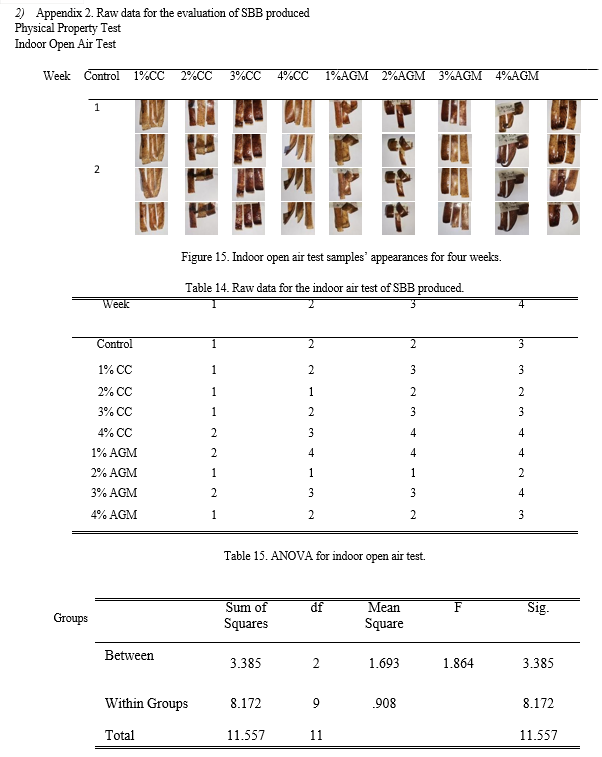
a. Indoor Open Air Test
The bioplastic produced with 2% CC and AGM has no noticeable changes in its appearances nor curled at all. On one hand, samples with 1% AGM are highly curled changing its orientation throughout the weeks. It also shows that the control sample have slightly curled throughout the month, minimum curling up starts to show in the second week. Analysis of variance employed revealed that the significance probability or p-value is equal to 0.210 which is greater than 0.05. This means that there is no significant difference among the groups in terms of indoor open air test.
Table 5. Characterization of indoor open air test samples’ appearances.
|
|
WEIGHTED MEAN |
DESCRIPTIVE INTERPRETATION |
|
Control |
2.00 |
Slightly curled |
|
1% CC |
2.25 |
Slightly curled |
|
2% CC |
1.50 |
No changes |
|
3% CC |
2.25 |
Slightly curled |
|
4% CC |
3.25 |
Moderately curled |
|
1% AGM |
3.50 |
Highly curled |
|
2% AGM |
1.25 |
No changes |
|
3% AGM |
3.00 |
Moderately curled |
|
4% AGM |
2.00 |
Slightly curled |
|
Legend: 4.20-5.00 - |
Completely curled |
|
3.40-4.19 - Highly curled 2.50-3.39 – Moderately curled 1.80-2.49 – Slightly curled 1.00-1.79 - No changes
b. Outdoor Open Air Test
The table 6 shows that the 2% AGM exhibits little to no changes in the duration of four weeks. Based on the interpretation, samples with 1% CC, 4% CC, and 3% AGM highly curled up while 1% AGM curled completely. It is recorded that the samples expanded over time due to moisture it may absorbed in the environment. The p-value calculated using ANOVA is 0.229 which is higher than the alpha value of 0.05 making the groups not significantly different with each other.
Table 6. Characterization of outdoor open air test samples’ appearances.
|
|
WEIGHTED MEAN |
DESCRIPTIVE INTERPRETATION |
|
Control |
2.00 |
Slightly curled |
|
1% CC |
3.50 |
Highly curled |
|
2% CC |
2.25 |
Slightly curled |
|
3% CC |
3.25 |
Moderately curled |
|
4% CC |
4.00 |
Highly curled |
|
1% AGM |
4.25 |
Completely curled |
|
2% AGM |
1.50 |
No changes |
|
3% AGM |
4.00 |
Highly curled |
|
4% AGM |
3.25 |
Moderately curled |
|
Legend: 4.20-5.00 - |
Completely curled |
|
3.40-4.19 - Highly curled 2.50-3.39 – Moderately curled 1.80-2.49 – Slightly curled 1.00-1.79 - No changes
2. Chemical Properties Test
Biodegradability test, strong and weak acid test, strong and weak base test, organic solvent test, water vapor transmission rate test, water absorption test and flammability test were used to determine the chemical properties of the SBB.
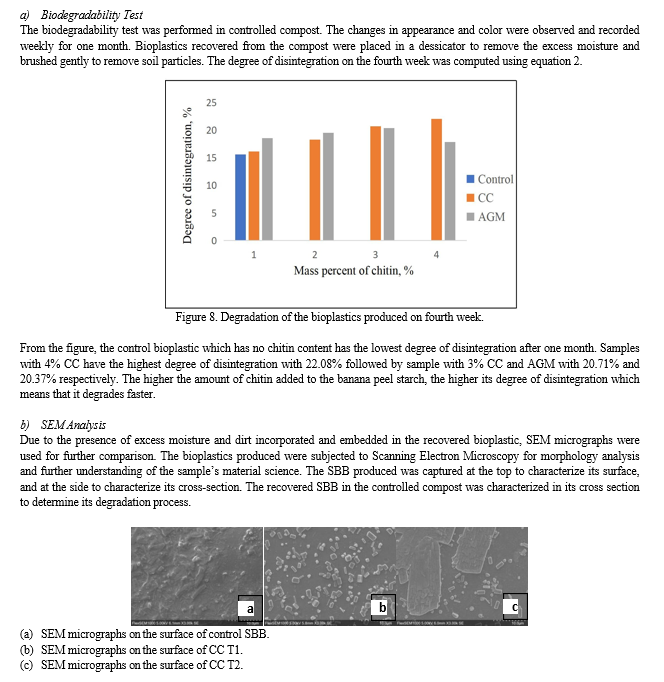


After four weeks burried under the controlled compost, the control SBB exhibits bulging and minimum signs of degradation. Its cross-section remains firm and intact, exhibiting slow degradation compared to the other samples. For the SBB produced with CC, films break down by expansion just like the control, only there is a wider area of degraded parts. On one hand, the SBB produced with AGM exhibits faster degradation, making it more superior. The micrographs show that the samples degrade through the holes and breakages in their cross-section. The best sample in terms of biodegradability tests is the 3% AGM.
Since the BPS were soaked in sodium metabisulphite solution which is a preservative and an anti-oxidant, it may be a factor that have affected the degradation of the bioplastic, improving its shelf-life (Ahmed, 2017 cited by Fabillar, 2019).
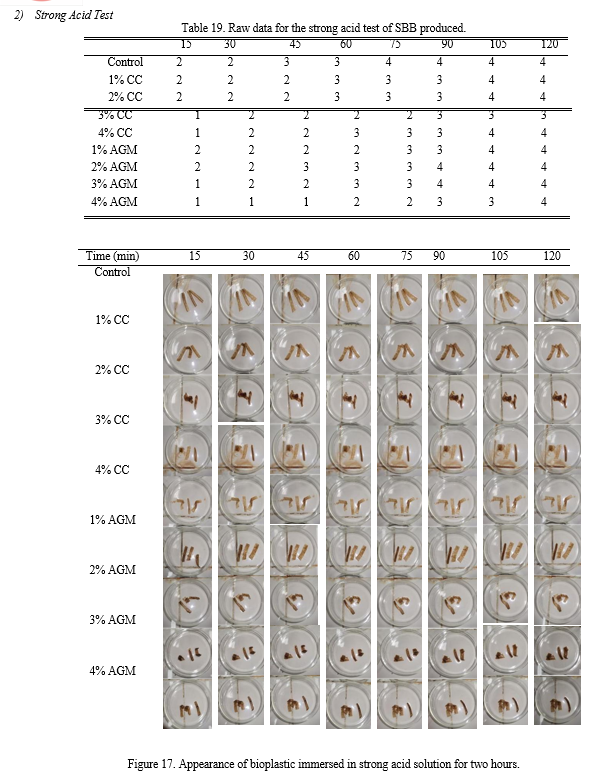
c. Acid Test
The bioplastics were tested by immersing it to strong and weak acid solution; 1M of Sulfuric acid was used for the strong acid test while 1M of Acetic acid was used for the weak acid test. The changes in appearance and color were observed and recorded every 15 minutes bracket with duration of two hours. Likert scale was used to quantify and characterize the appearance of the bioplastics.
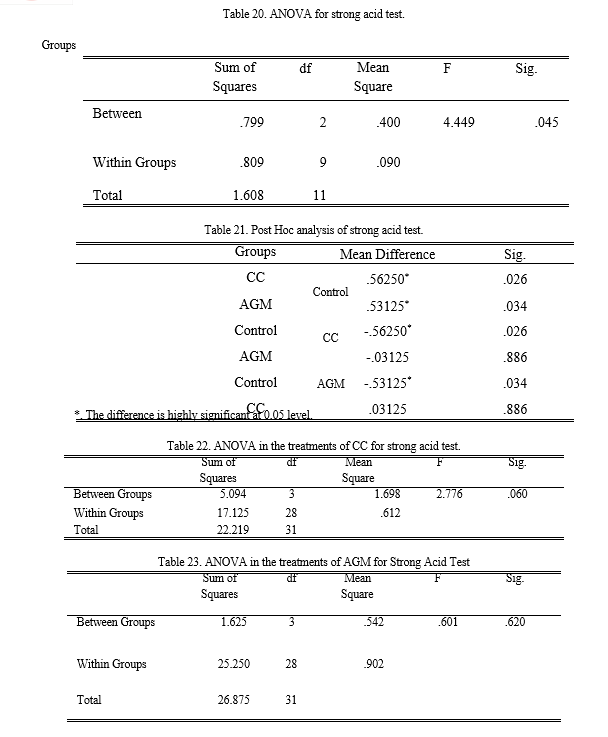
Based from Table 7, the samples with 3% CC and 4% AGM slightly dissolved in the strong acid solution while other treatments fall in the same category which is moderately dissolved. Analysis of variance employed revealed that there is a significant difference between the control, CC, and AGM group with its 0.045 p- value. Fisher’s Least Significant Difference (LSD) Post Hoc test was employed to determine differences. Results showed that the SBB produced with CC and AGM is superior interms of stability in strong acid compared to the control. However, CC and AGM exhibits no significant difference with each other making them have the same effectiveness in this test.
Table 7. Characterization of strong acid test samples’ appearances.
|
|
WEIGHTED MEAN |
DESCRIPTIVE INTERPRETATION |
|
Control |
3.25 |
Moderately dissolved |
|
1% CC |
2.875 |
Moderately dissolved |
|
2% CC |
2.875 |
Moderately dissolved |
|
3% CC |
2.25 |
Slightly dissolved |
|
4% CC |
2.75 |
Moderately dissolved |
|
1% AGM |
2.75 |
Moderately dissolved |
|
2% AGM |
3.125 |
Moderately dissolved |
|
3% AGM |
2.875 |
Moderately dissolved |
|
4% AGM |
2.125 |
Slightly dissolved |
|
Composite Mean |
2.7639 |
Moderately dissolved |
|
Legend: 4.20-5.00 - 3.40-4.19 - |
Completely dissolved (CD) Highly dissolved (HD) |
|
2.50-3.39 – Moderately dissolved (MD) 1.80-2.49 – Slightly dissolved (SD) 1.00-1.79 - No changes (NC)
The treatments of each group also undergone another ANOVA testing to determine the best concentration of chitin. The results of the analysis showed that there are no significant difference between the treatments of either CC and AGM group.
On one hand, the results of the weak acid test based from Table 8 shows that the samples with 2% CC and 2% AGM moderately dissolved in the weak acid solution while all of the other treatments were slightly dissolved. The p-value calculated using ANOVA is 0.969 making the groups not significantly different with each other.
Table 8. Characterization of weak acid test samples’ appearances.
|
|
WEIGHTED MEAN |
DESCRIPTIVE INTERPRETATION |
|
Control |
2.375 |
Slightly dissolved |
|
1% CC |
1.875 |
Slightly dissolved |
|
2% CC |
3.125 |
Moderately dissolved |
|
3% CC |
2.375 |
Slightly dissolved |
|
4% CC |
2.375 |
Slightly dissolved |
|
1% AGM |
2.375 |
Slightly dissolved |
|
2% AGM |
3.125 |
Moderately dissolved |
|
3% AGM |
2.125 |
Slightly dissolved |
|
4% AGM |
2.125 |
Slightly dissolved |
|
Composite Mean |
2.4306 |
Slightly dissolved |
|
Legend: 4.20-5.00 - |
Completely dissolved (CD) |
|
3.40-4.19 - Highly dissolved (HD) 2.50-3.39 – Moderately dissolved (MD) 1.80-2.49 – Slightly dissolved (SD) 1.00-1.79 - No changes (NC)
d. Base Test
The bioplastics were tested by immersing it to strong and weak base solution; 1M of Sodium hydroxide was used for the strong base test while 1M of Ammonium hydroxide was used for the weak base test.
The changes in appearance and color were observed and recorded every 15 minutes bracket with duration of two hours. Likert scale was used to quantify and characterize the appearance of the bioplastics.
Table 9. Characterization of strong base test samples’ appearances.
|
|
WEIGHTED MEAN |
DESCRIPTIVE INTERPRETATION |
|
Control |
1.75 |
No changes |
|
1% CC |
2.00 |
Slightly dissolved |
|
2% CC |
2.125 |
Slightly dissolved |
|
3% CC |
2.125 |
Slightly dissolved |
|
4% CC |
1.875 |
Slightly dissolved |
|
1% AGM |
2.00 |
Slightly dissolved |
|
2% AGM |
2.375 |
Slightly dissolved |
|
3% AGM |
1.75 |
No changes |
|
4% AGM |
1.75 |
No changes |
|
Composite Mean |
1.9722 |
Slightly dissolved |
|
Legend: 4.20-5.00 - |
Completely dissolved (CD) |
|
3.40-4.19 - Highly dissolved (HD) 2.50-3.39 – Moderately dissolved (MD) 1.80-2.49 – Slightly dissolved (SD) 1.00-1.79 - No changes (NC)
Based from Table 9, the control sample and that with 3% AGM and 4% AGM showed no changes in the duration of two hours. While all the other treatments slightly dissolved in the strong base solution. The color starts to diffuse around 30 minutes prior to immersing it to the solution. The p-value calculated using ANOVA is 0.130 making the groups not significantly different with each other.
Table 10. Characterization of weak base test samples’ appearances.
|
|
WEIGHTED MEAN |
DESCRIPTIVE INTERPRETATION |
|
Control |
2.625 |
Moderately dissolved |
|
1% CC |
2.375 |
Slightly dissolved |
|
2% CC |
2.625 |
Moderately dissolved |
|
3% CC |
2.875 |
Moderately dissolved |
|
4% CC |
2.625 |
Moderately dissolved |
|
1% AGM |
2.375 |
Slightly dissolved |
|
2% AGM |
2.625 |
Moderately dissolved |
|
3% AGM |
2.25 |
Slightly dissolved |
|
4% AGM |
2.50 |
Moderately dissolved |
|
Composite Mean |
2.5417 |
Moderately dissolved |
|
Legend: 4.20-5.00 - |
Completely dissolved (CD) |
|
3.40-4.19 - Highly dissolved (HD) 2.50-3.39 – Moderately dissolved (MD) 1.80-2.49 – Slightly dissolved (SD) 1.00-1.79 - No changes (NC)
Based from Table 10, the samples with 1% CC, 1% AGM and 3% AGM slightly dissolved while all the other treatments moderately dissolved in the weak base solution. Unlike the strong base test, the color of the sample did not diffuse in the solution. The p-value calculated using ANOVA is 0.181 making the groups not significantly different with each other.
e. Organic Solvent Test
The bioplastics were tested by immersing it to 20% (v/v) Ethanol. The changes in appearance and color were observed and recorded every 15 minutes bracket with duration of two hours. Likert scale was used to quantify and characterize the appearance of the bioplastics.
Table 11. Characterization of organic solvent test samples’ appearances.
|
|
WEIGHTED MEAN |
DESCRIPTIVE INTERPRETATION |
|
Control |
2.75 |
Moderately dissolved |
|
1% CC |
3 |
Moderately dissolved |
|
2% CC |
2.75 |
Moderately dissolved |
|
3% CC |
3.25 |
Moderately dissolved |
|
4% CC |
2.75 |
Moderately dissolved |
|
1% AGM |
2.625 |
Moderately dissolved |
|
2% AGM |
2.875 |
Moderately dissolved |
|
3% AGM |
2.125 |
Slightly dissolved |
|
4% AGM |
2.5 |
Moderately dissolved |
|
Composite Mean |
2.7361 |
Moderately dissolved |
|
Legend: 4.20-5.00 - |
Completely dissolved (CD) |
|
3.40-4.19 - Highly dissolved (HD) 2.50-3.39 – Moderately dissolved (MD) 1.80-2.49 – Slightly dissolved (SD) 1.00-1.79 - No changes (NC)
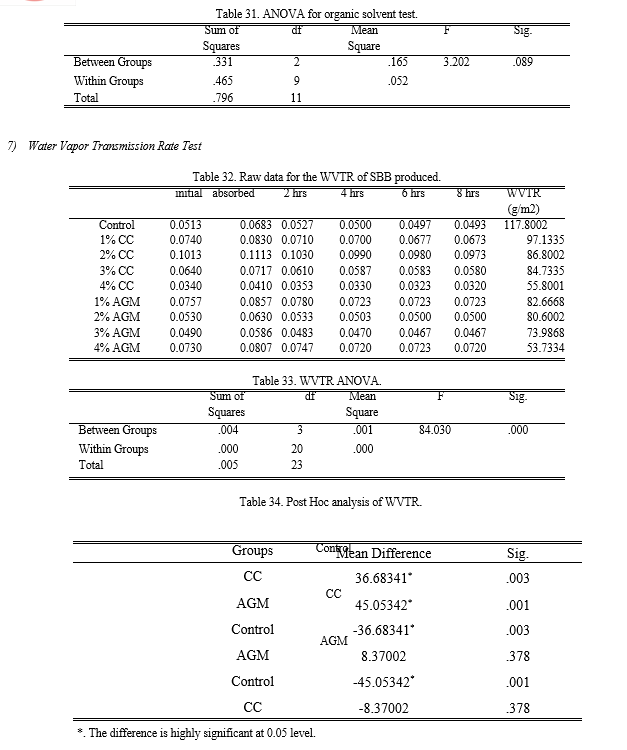
Based from Table 11, the samples with 3% AGM slightly dissolved while all the other treatments moderately dissolved in the ethanol solution. The p-value calculated using ANOVA is 0.089 making the groups not significantly different with each other.
f. Water Vapor Transmission Rate Test
The WVTR of the bioplastics was tested to measure and compare their ability to resist moisture transmission. The bioplastic samples were evaluated every 2 hours for 8 hours to determine the transformation mass. The desirable effect of chitin in the SBB produced was to limit the transmission rate of moisture so that it can be an effective packaging.
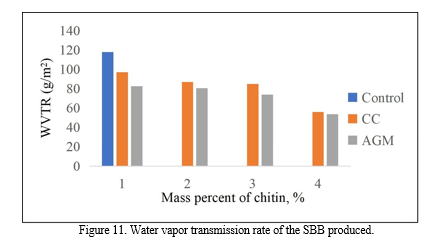
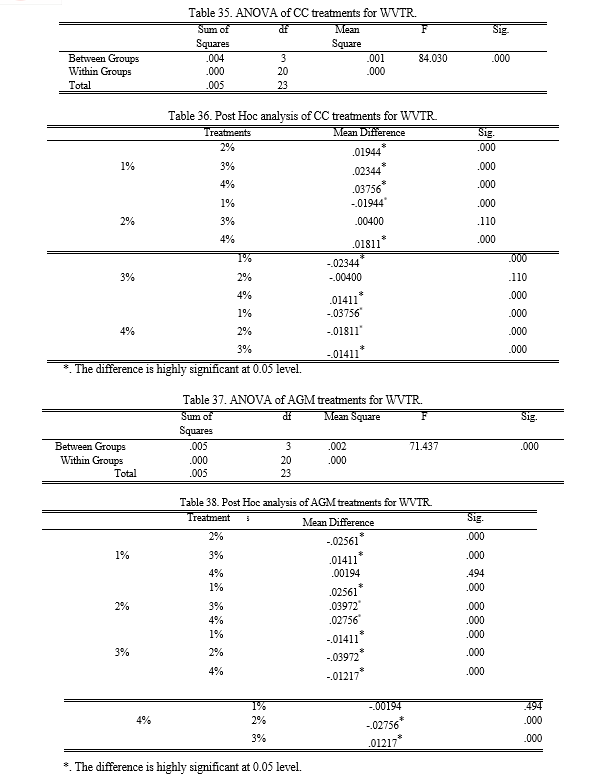
Since, WVTR is the ability to resist moisture transmission; the lower values indicate the better moisture protection. The control recorded the highest WVTR which is 117. 8 g/m2 while both the 4% CC and AGM recorded the lowest with 55.8 g/m2 and 53.73 g/m2 respectively. Figure 14 also shows that the higher the amount of chitin added to the banana peel starch, the bioplastic produce has a better resistance to moisture. The results of the ANOVA employed revealed that the p-value is equal to 0.002 making the groups have significant difference with each other. Through Fisher’s LSD Post Hoc analysis, it is determined that SBB incorporated with CC and AGM has the same effectiveness and is better compared to the control. Through separate ANOVA and Post Hoc analysis, it showed that the optimum results for the WVTR from CC is T4 which is addition of 4% chitin. While T3 is for the AGM since the 3% and 4% extracted chitin addition has no significant difference.
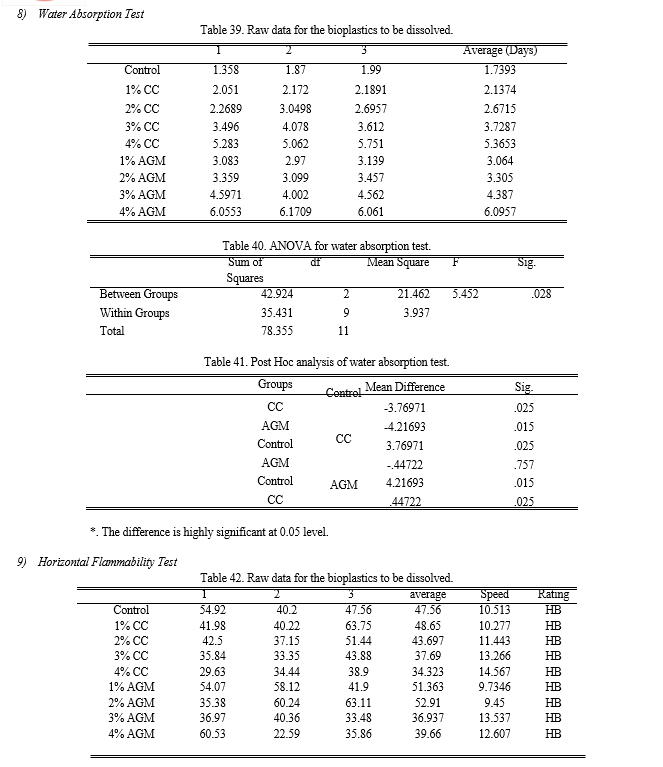
g. Water Absorption Test
The samples from the different treatments were immersed in distilled water and the time required for them to dissolve was recorded. All the samples were dissolved within 10 days, wherein the fastest time recorded for a sample to be dissolved was around 2 days. It is expected that the chitin addition will improved the water-barrier properties of the SBB produced resulting to higher number of solubility period.
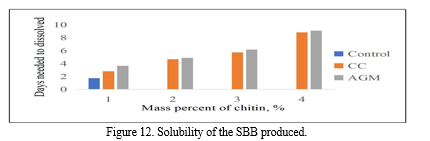
Figure 12 shows that the control bioplastic has the fastest solubility with an average of 1.739 days. While bioplastic containing 4% chitin of AGM and CC had the slowest solubility with an average of 8.832 days and 9.096 days respectively. From the results, the higher the mass of chitin added to the banana peel starch, the produced bioplastics took longer to break down and dissolve in distilled water. The results of the ANOVA employed revealed that the p-value is equal to 0.028 making the groups significantly different with each other. Through Fisher’s LSD Post Hoc analysis, it is determined that SBB incorporated with CC and AGM has the same effectiveness and is better compared to the control. Through separate ANOVA and Post Hoc analysis, it showed that the optimum results was obtained from both the T4 of CC and AGM.
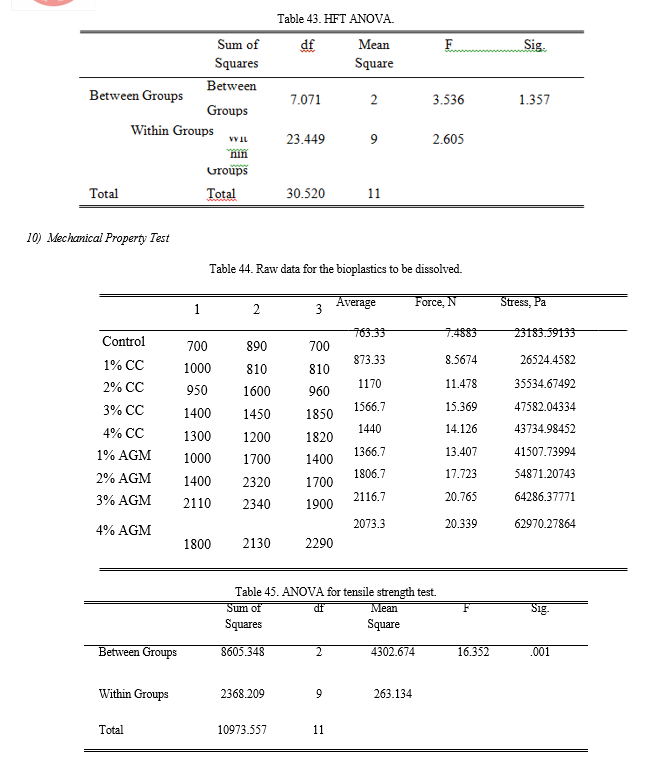
h. Flammability Test
The samples were tested while in horizontal position by igniting one end and recording the time needed for the flame to reach the other end of the sample. All the samples tested were flammable and were all burned after just one ignition.
The results of the flammability rate of the bioplastics are shown in Figure 16. The fastest flammability rate recorded was 14.5673 mm2/s which was exhibited by the bioplastic with 4% CC. Followed by the 3% AGM with a rate of 13.5367 mm2/s, and the slowest was the 2% AGM with a rate of 9.45 mm2/s. The p-value calculated using ANOVA is 0.305 making the groups not significantly different with each other.
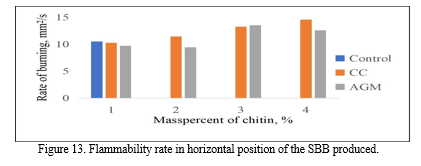
On one hand, the vertical flammability test of the samples was performed with the use of a candle wherein the time taken for the samples to drip was recorded. According to the standards made by Underwriters Laboratory, vertical flammability test is mainly concern on the dripping of the material wherein a good material should not produce a drip if possible. Since all the treatments of the SBB produced does not produce any drip, it can be considered as a good bioplastic.
h. Tensile Strength Test
The tensile strength test was done with an improvised set-up in which one end of the sample was attached to a support while the other end was attached to the spring balance. The weight of the weights needed to tear the samples was recorded. It was found out that the bioplastics from incorporated with commercialize chitin carried a weight range of 810 g to 1850 g that was approximately equivalent to a tensile strength of 24,600 Pa to 56,200 Pa. The bioplastic incorporated with extracted chitin from AGM carried a weight range of 1000 g to 2340 g that was approximately equivalent to a tensile strength of 30,300 Pa to 71,000 Pa.
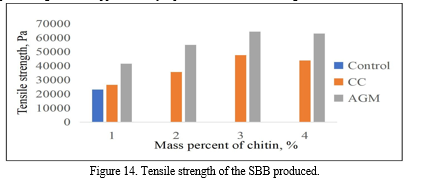
The results of the ANOVA employed revealed that the p-value is equal to 0.020 making the groups significantly different with each other. Through Fisher’s LSD Post Hoc analysis, it is determined that SBB incorporated with CC is better than the control but is inferior compared to the AGM. Through separate analysis, it is concluded that the treatments under AGM has no significant difference, it means that using T1 can still have the same effectiveness in that of T2, T3, and T4 interms of tensile stress test.






Conclusion
A. Summary The saba banana peels were collected from public markets and residential waste. The shells for chitin extraction were gathered from seafood restaurants, residential food waste, and rice fields in Brgy. 8, San Antonio, Sarrat, Ilocos Norte. While the commercialized chitin labeled as technical grade chitin extracted from shrimp shell was purchased in Amazon. The AGM and GASS were extracted to produce chitin through deproteinization and demineralization extraction process. Extraction was carried out through deproteinization using 2M NaOH with a ratio of 20ml : 1g (v/w) of NaOH and demineralization using 2M HCL with a ratio of 20ml : 1g (v/w) of HCL. Based from the percentage extracted, the AGM has a greater yield of chitin than the GASS which makes it the CCE to be used in the production of bioplastic. Four treatments were done using the amounts of chitin in 1.0 %, 2.0 %, 3.0 % and 4.0 % by wt. The amount of BPS, PVA solution, glycerol, and distilled water were constant for all the treatments. Three replicates for each treatment were used for a total of 459 sample strips. The strips were used for the different tests which included SEM, air test, biodegradability test, chemical tests, water vapor transmission rate and absorption test, flammability test and tensile stress test. Analysis of Variance (ANOVA) was employed to analyze the data gathered and determine the best treatment for the different tests involved. Based from findings, the bioplastic produced from AGM and CC were superior in terms of strong acid test, water vapor transmission rate test, water absoption test, and tensile stress test than to the control. On the other tests, chitin addition has no significant difference in the properties of SBB. In terms of strong acid and water absorption test, CC and AGM exhibit the same effectiveness, and the concentration of chitin does not affect its properties. In water vapor transmission rate test, CC and AGM have no significant difference and it showed that the best treatment for CC is T4 and that for AGM is T3. In the tensile stress test, AGM is found to be better than CC, and its treatments have no significant difference with each other. Overall, this study proves that AGM was a good alternative for CC interms of reinforcing water barrier and tensile strength properties of the SBB. B. Conclusion Bioplastic is possible to be produced using the chitin of AGM and BPS mixed to a constant amount of PVA solution (100ml) and glycerol (2.5ml). The AGM was a good alternative for CC and is ideal for packaging. Since AGM is invasive and considered as food waste, it makes it an economical choice as raw material for bioplastic. The SBB incorporated with AGM and CC was more stable than the Control in terms of strong acid test, water vapor transmission rate test, water absoption test, and tensile stress test in which chitin from AGM optimized the bioplastic. Comparing the best treatment, it was found that the best treatment overall was the SBB produced incorporated with 3% AGM. C. Recommendations Based from the results and data gathered by the researcher, the following are being recommended: 1) Conduct study on extraction of chitin using various methods 2) Optimize different concentrations of AGM and GASS 3) Use of other raw materials as substitute for chitin source 4) Study other renewable resources as a source for the production of bioplastic 5) Use a bleaching agent to improve the appearance of bioplastics produced from banana peel and use anti-oxidants to improve the transparency of the bioplastic produced 6) Use of coating to enhance the stability of bioplastics when in contact with moist or wet object or use of another polymer which is water-proof but is biodegradable 7) Use different plasticizers and investigate how the different properties of the produced bioplastic will be affected 8) Investigate different proportions of raw materials, polymer and plasticizer that can produce edible bioplastic 9) Conduct stability study on the different parameters after a certain period of time
References
[1] Abdulkarim, A., Isa, M. T., Abdulsalam, S., Muhammad, A. J., and Ameh, A. O. (2013). Extraction and Characterisation of Chitin and Chitosan from. Civil and Environmental Research, 3(2), 108–115. [2] Agsalog, C. (2017). Production of biodegradable plastic produced from different types of waste paper (white paper, yellow paper and newspaper) as cellulose source. -College of Engineering, Mariano Marcos State University, City of Batac. [3] Atiwesh, G., Mikhael, A., Parrish, C. C., Banoub, J., and Le, T. A. T. (2021). Environmental impact of bioplastic use: A review. Heliyon, 7(9), e07918. https://doi.org/10.1016/j.heliyon.2021.e07918 [4] British Plastics Federation (2022). “The benefits of Plastics Materials.” Retrieved from https://www.bpf.co.uk [5] Boukhlifi, F. (2019). Sustainable treatment of heavy metals by absorption on rawchitin/chitosan. DOI: 10.5772/intechopen.88998 [6] Cabacungan, K. (2006). Comparative study of biodegradable plastic produced using cadena de amor, cassava, sweet potato and taro as an additive. College of Engineering, Mariano Marcos State University, City of Batac. [7] Cadano, J. R., Jose, M., Lubi, A. G., Maling, J. N., Moraga, J. S., Shi, Q. Y., Vegafria, H. M., and VinceCruz-Abeledo, C. C. (2021). A comparative study on the raw chitin and chitosan yields of common bio-waste from Philippine seafood. Environmental Science and Pollution Research, 28(10), 11954–11961. https://doi.org/10.1007/s11356-020-08380-5 [8] Chandarana, J. and Sai Chandra, P. (2021). Production of bioplastics from banana peels. International Journal of Scientific Research & Engineering Trends, Vol. 7 No. 1, Anurag Group of Institutions, Ghatkesar, Hyderabad, India. [9] Cho, R. (2017). The truth about bioplastics. Colombia Climate School. Retrieved from https://news.climate.columbia.edu/2017/12/13/the-truth-about-bioplastics/ [10] da Silva Júnior, W. F., de Oliveira Pinheiro, J. G., Moreira, C. D. L. F. A., de Souza, F. J. J., and de Lima, Á. A. N. (2017). Alternative Technologies to Improve Solubility and Stability of Poorly Water-Soluble Drugs. In Multifunctional Systems for Combined Delivery, Biosensing and Diagnostics. Elsevier Inc. https://doi.org/10.1016/B978-0-323-52725-5.00015-0 [11] Dawam Abdullah, A. H., Fikriyyah, A. K., and Furghoniyyah, U. (2020). Effect of chitin addition on water resistance properties of starch-based bioplastic properties. IOPConference Series: Earth and Environmental Science, 483(1). https://doi.org/10.1088/1755-1315/483/1/012002 [12] Delgado, J. M. P. Q., Da Silva, V., Nasser, R. O., Gonçalves, M. P., and Andrade, C. T. (2009). Water sorption isotherms and textural properties of biodegradable starch-based superabsorbent polymers. Defect and Diffusion Forum, 283–286, 565–570. https://doi.org/10.4028/www.scientific.net/DDF.283-286.565 [13] Dragan, E. S., Humelnicu, D., and Dinu, M. V. (2019). Development of chitosan- poly(ethyleneimine) based double network cryogels and their application as superadsorbents for phosphate. Carbohydrate Polymers, 210 (November 2018), 17–25. https://doi.org/10.1016/j.carbpol.2019.01.054 [14] Edaes, F. and de Souza, C. (2020). Conventional plastics\' harmful effects and biological and molecular strategies for biodegradable plastics\' production. Current Biotechnology, Vol. 9 No. 4. [15] Elieh-Ali-Komi, D. and Hamblin, M. (2016). Chitin and Chitosan: Production and application of versatile biomedical nanomaterials. Retrieved from https://pubmed.ncbi.nlm.nih.gov/27819009/ [16] Fabillar, M. (2019). Characterization of bioplastics properties produced from banana peel and seaweed gracilaria. College of Engineering, Mariano Marcos State University. [17] Fathanah, U., Lubis, M. R., Nasution, F., and Masyawi, M. S. (2018). Characterization of bioplastic based from cassava crisp home industrial waste incorporated with chitosan and liquid smoke. IOP Conference Series: Materials Science and Engineering, 334(1). Retrieved from https://doi.org/10.1088/1757- 899X/334/1/012073 [18] Gadhave, R., Das, A., Mahanwar, P. and Gadekar, P. (2018). Starch based bio- plastics: the future of sustainable packaging.Open J. Polym. Chem.2018,8, 21– 33. [19] Gibbens, S. (2018). “What you need to know about plant-based plastics.” Retrieved from https://www.nationalgeographic.com/environment/article/are-bioplastics-made-from-plants-better-for-environment-ocean-plastic. [20] Gaonkar. M.R, Palaskar. P, N. R. (2017). Production of bioplastic from banana peels. I, 6(1), 526–536. [21] Havstad, M. (2020). Plastic Waste and Recycling, Chapter 5- Biodegradable plastics. Retrieved from https://doi.org/10.1016/B978-0-12-817880-5.00005-0 [22] Hongpattarakere, T. and Uraipan, S. (2014). Bifidogenic characteristic and protective effect of saba starch on survival of Lactobacillus plantarum CIF17AN2 during vaccuum-drying and storage. Carbohydrate polymers 117, pp. 255-261. Retrieved from DOI: 10.1016/j.carbpol.2014.09.065 [23] Huzaisham, N. A., and Marsi, N. (2020). Utilization of banana (Musa paradisiaca) peel as bioplastic for planting bag application. International Journal of Advanced Research in Engineering and Technology, 11(4), 108–118. https://doi.org/10.34218/IJARET.11.4.2020.013 [24] Jayakumar, R., Prabaharan, M., Nair, S. V., and Tamura, H. (2010). Novel chitin and chitosan nanofibers in biomedical applications. Biotechnology Advances, 28(1), 142–150. https://doi.org/10.1016/j.biotechadv.2009.11.001 [25] Jiang, T., Duan, Q., Zhu, J., Liu, H., and Yu, L. (2020). Starch-based biodegradable materials: Challenges and opportunities. Advanced Industrial and Engineering Polymer Research, 3(1), 8–18. Retrieved from https://doi.org/10. 1016/j.aiepr.2019.11.003 [26] Judawisastra, H. et al. (2017). Water absorption and its effect on the tensile properties of tapioca starch/polyvinyl alcohol bioplastics. IOP Conf. Ser.: Mater. Sci. Eng.223012066 [27] Kaewboonruang, S., Phatrabuddha, N., Sawangwong, P., and Pitaksanurat, S. (2016). Comparative Studies on the Extraction of Chitin – Chitosan from Golden Apple Snail Shells at the Control Field. IOSR Journal of Polymer and Textile Engineering (IOSR-JPTE), 3(1), 34–41. https://doi.org/10.9790/019X-03013441 [28] Manimaran, D. S., Nadaraja, K. R., Vellu, J. P., Francisco, V., Kanesen, K., and Yusoff, Z. Bin. (2020). Production of Biodegradable Plastic from Banana Peel. International Journal of Innovative Research in Science, Engineering and Technology, 9(7), 6177. www.ijirset.com [29] Marichelvam, M. K., Jawaid, M., and Asim, M. (2019). Corn and rice starch-based bio-plastics as alternative packaging materials. Fibers, 7(4), 1–14. https://doi.org/10.3390/fib7040032 [30] Miyata, Y. (1994). Biodegradable Plastics. In Nippon Nogeikagaku Kaishi (Vol. 68, Issue 9). https://doi.org/10.1271/nogeikagaku1924.68.1318 [31] Natarajan, T., N, K., and S, R. (2017). Extraction and Characterization of Chitin and Chitosan from Achatinodes. Natural Products Chemistry & Research, 05(06). https://doi.org/10.4172/2329-6836.1000281 [32] Oyekunle, D. T., and Omoleye, J. A. (2019). Extraction, characterization and kinetics of demineralization of chitin produced from snail shells of different particle sizes using 1.2 M HCl. International Journal of Mechanical Engineering and Technology, 10(1), 2011–2020. [33] Palaniveloo, K., Amran, M. A., Norhashim, N. A., Mohamad-Fauzi, N., Peng- Hui, F., Hui-Wen, L., Kai-Lin, Y., Jiale, L., Chian-Yee, M. G., Jing-Yi, L., Gunasekaran, B., and Razak, S. A. (2020). Food waste composting and microbial community structure profiling. Processes, 8(6), 1–30. https://doi.org/10.3390/pr8060723 [34] Parker, L. (2018). “Here’s how much plastic trash littering the earth.” Retrieved from https://www.nationalgeographic.com/science/article/plastic-produced- recycling-waste-ocean-trash-debris-environment. Perna viridis. Retrieved from https://www.cabi.org/isc/datasheet/70090. [35] Rivadeneira, J. P., Wu, T., Ybanez, Q., Dorado, A. A., Migo, V. P., Nayve, F. R. P., and Castillo-Israel, K. A. T. (2020). Microwave-assisted extraction of pectin from “Saba” banana peel waste: Optimization, characterization, and rheology study. International Journal of Food Science, 2020. https://doi.org/10.1155/2020/8879425 [36] Rizwana Beevi, K., Sameera Fathima, A. R., Thahira Fathima, A. I., Thameemunisa, N., Noorjahan, C. M., and Deepika, T. (2020). Bioplastic synthesis using banana peels and potato starch and characterization. International Journal of Scientific and Technology Research, 9(1), 1809–1814. [37] Rios, L., Moore, C. and Jones, P. (2007). Persistent organic pollutants carried by synthetic polymers in the ocean environment. Marine Pollution Bulletin 54:1230–1237. [38] Robertson, J. (2004). 11119. The American Mathematical Monthly, 111(10), 915. https://doi.org/10.2307/4145104 [39] Suman, T. Y., Li, W. G., Alif, S., Faris, V. R. P., Amarnath, D. J., Ma, J. G., and Pei, D. S. (2020). Characterization of petroleum-based plastics and their absorbed trace metals from the sediments of the Marina Beach in Chennai, India. Environmental Sciences Europe, 32(1). https://doi.org/10.1186/s12302-020- 00388-5 [40] Swapp, S. (2017). Scanning Electron Microscopy (SEM). Retrieved from https://serc.carleton.edu/research_education/geochemsheets/techniques/SEM.htm l [41] Tiseo, I. (2021). Gobal plastic production 1950-2020. Retrieved from https://www.statista.com/statistics/282732/global-production-of-plastics-since- 1950/. [42] Yin, P., Liu, J., Zhou, W., and Li, P. (2020). Preparation and Properties of Corn Starch / Chitin. Polymers. [43] Younes, I., and Rinaudo, M. (2015). Chitin and chitosan preparation from marine sources. Structure, properties and applications. Marine Drugs, 13(3), 1133–1174. https://doi.org/10.3390/md13031133 [44] Wagner, C. (2014). “Types, applications, toxicity and regulation of bioplastics used in food contact materials.” Retrieved from https://www.foodpackagingforum.org/food-packaging-health/bioplastics
Copyright
Copyright © 2023 Mark Khevin L. De Guzman, April Jieren R. Muska. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET55639
Publish Date : 2023-09-06
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here

